A Non-alternant Aromatic Belt: Methylene-bridged [6]Cycloparaphenylene Synthesized from Pillar[6]arene
Yuanming Li, Yasutomo Segawa, Akiko Yagi, Kenichiro Itami
J. Am. Chem. Soc. 2020, accepted. DOI:10.1021/jacs.0c06007
我々は、メチレン架橋された[6]CPPである”non-alternant aromatic belt (五員環含有ベルト)”の合成に成功した。
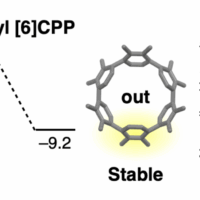
フェニレン上にトリフレート基を有するピラー[6]アレーンに対し、ニッケル錯体を用いたアリール-アリール分子内カップリング反応を行うことによって、18%の単離収率で五員環含有ベルトを得た。[6]CPPをメチレン架橋することにより、隣接するフェニレンユニット間のπ共役が拡張されるとともにエネルギーギャップが小さくなることがわかった。さらに、五員環含有ベルトの環内部に小分子を包摂することで、超分子構造体が形成されることを明らかにした。
The synthesis, structure, and properties of methylene-bridged [6]cycloparaphenylene ([6]CPP), a non-alternant aromatic belt, are described. This belt-shaped methylene-bridged [6]CPP, in which each phenylene unit is tethered to its neighbors by methylene bridges, was constructed through six-fold intramolecular nickel-mediated aryl-aryl coupling of triflate-functionalized pillar[6]arene in 18% isolated yield. Compared with the analogous [6]CPP, the methylene bridges co-planarize neighboring paraphenylene units and enhance the degree of pi-conjugation, resulting in a significant decrease in energy gap. Moreover, the incorporation of small molecules in the defined pocket of methylene-bridged [6]CPP makes it an attractive supramolecular architecture. Methylene-bridged [6]CPP is characterized by high internal strain energy reaching 110.2 kcal·mol–1, attributed to its restricted structure. This work not only exhibits an efficient strategy to construct a new family of aromatic belt, but also showcases their properties, which combine the merits of CPPs and pillararenes.


 English
English